- November 29, 2021
Electroplating is an important and indispensable link in the contemporary industrial chain and is widely used in numerous industries such as machinery, electronics, automobiles, aviation, and aerospace, etc. It plays a role in decorating, protecting, and increasing the functionality of various industrial products to enable them to meet various practical needs.
What is Electroplating?

How to definition electroplating? Electroplating is one of the post finishing processes from prototype design to mass production. By plating, a product can be given a beautiful appearance and at the same time provide corrosion protection.
Electroplating is also called electrodeposition, and the five basic hardware for the process are a DC power supply, a plating bath, a plating solution containing ions of the plated metal, anodes (the metal to be plated layer), and cathodes (the workpiece to be processed), and conductive rods, cables, and hangers. By connecting these five pieces of hardware to form a conductive circuit, the accumulation of plating ions on the surface of the processed part is achieved through the transfer of electrons, thus reducing it to a metal coating.
How does Electroplating work?
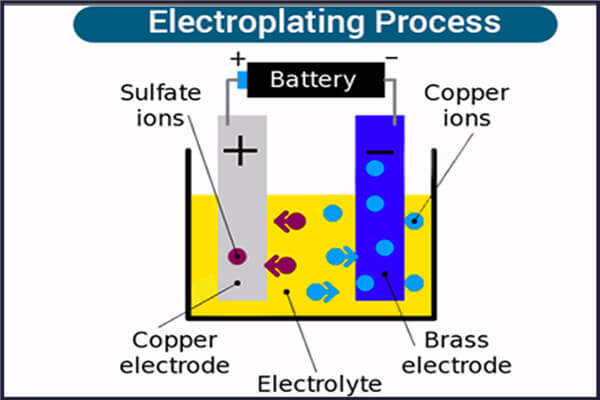
The electroplating process is an application of an electrolytic cell that requires two different metals, an electrolyte solution, two electrodes, and a battery or other power source that generates an electric current.
To begin, determine which anode and cathode and electrolyte to use by determining which chemical reaction or reactions you want to occur when the electric current is turned on.
The next step is to make sure that the electrodes you’re going to plate is entirely clean. Instead, when metal atoms from the electrolyte are deposited on it, they will not establish a strong bond and would rub off. Cleaning is usually accomplished by putting the electrodes in an acid solution or acidic mixture or (briefly) reversing the electroplating circuit. If the electrode is exceptionally clean, atoms from the plating metal form a strong link with it by adhering to the crystallographic craft’s exterior borders.
A positive ion is an atom with too few electrons. When they reach the cathode, they combine with electrons and lose their positive charge. A negatively charged ion is an atom with too many electrons. When they reach the positive anode, they transfer electrons to the anode and lose their negative charge.
In a form of electroplating, the metal to be plated is at the anode of the circuit and the material to be plated is at the cathode. After a period of reaction, the positively charged metal molecules will slowly migrate to the negatively charged metal surface, forming a very thin layer, which is called the coating.
Materials Are Used In The Electroplating Process

There are many different types of metal materials suitable for post-plating.Including nickel, chromium, brass, cadmium, copper sulfate, gold, silver, tin, and zinc. Nickel and brass are commonly used. We can recommend suitable metal materials and electroplating solutions for your project according to your specific electroplating requirements.
Which plastic materials can be electroplated
In addition to metallic materials, many different plastics can be plated in this way, including ABS, phenolic plastics, urea-formaldehyde, nylon, and polycarbonate. You will often find electroplating plastic parts on automobiles, plumbing, household, and electrical fittings that appear to be metal but are actually plated plastic.
How are plastics electroplated?
As you know that thick plastic layers never conduct electricity, in practice, this implies we’ll need to treat our plastic differently to make it electrically conductible. There are various steps to this process. To begin, the plastic must be thoroughly cleaned to remove contaminants such as dust, filth, grease, and surface scratches. The surface is then etched with acid and treated with a catalyst (a chemical reaction accelerator) to ensure that a coating adheres to it. Let’s now dip it in a solution of copper sulfate or nickel to cover it with a thin layer of highly conductive metal. By applying these steps in appropriate way, plastic becomes electroplated such as metal.
Application of electroplating
Plating is an integral part of the manufacturing process and is used to improve corrosion resistance, wear resistance, and aesthetics. Plating is used in a wide range of industries and products, and there is a huge market demand for it. Here is a list of some of the industries that WayKen serves and how they use electroplating.
Automotive industrial

Electroplating is used in the automotive industry to help extend the life of a vehicle, improve its dynamics, and enhance its decorative properties. For example, for shock absorber linkages, chrome plating has a positive effect on ensuring the corrosion resistance and lubricity of the shock absorber linkage. Modern automobile decorative parts use an electroplating process, which has the role of heat vibration resistance, decoration, and corrosion resistance.
Electronic industry
Electroplating is an important means of anti-corrosion for electronic products, which can decorate the appearance and improve the texture and ornamental properties of products. In addition, electronic companies often use gold plating to improve electrical conductivity, applying it to semiconductors and connectors, functional plating is of great significance for electronic products.
Jewelry Industry
As society continues to develop and people’s aesthetic sense progresses, many types of plating available for jewelry plating have been developed in the modern gold and silver jewelry processing industry, such as gold plating, silver plating, platinum plating, rhodium plating, palladium plating, etc. for precious metal jewelry plating, in addition to rose gold plating, black gold plating, etc. Jewelry designers and manufacturers rely on this process to enhance the color, durability, and beauty of rings, bracelets, pendants, and various other items.
Medical industry
Electroplating is used in the medical industry to improve the functionality of various aspects of medical equipment. For example, aluminum electroplating, electroplating steel, and other metal can be used to conduct heat and electricity, provide good wear resistance, corrosion resistance, form strong welds, create antibacterial, biocompatible surfaces, and create markers and braided bundles of catheters that X-rays cannot penetrate.
Aerospace industry
The electroplating process is one of the powerful means to improve the surface properties of materials used in aerospace. Plating, a naturally smooth and thin finish, enhances the wear resistance of aerospace components. For example, zinc-nickel is the ability to reduce the effects of thermal stress on aircraft components, and the nickel plating layer and titanium alloy material have good bonding ability, which can improve the surface properties of titanium electroplating.
Oil and gas industry
The electroplating process can greatly protect oil and gas tools and other components from corrosion, thus improving overall performance and longevity. Applications of the plating process can be found in other areas such as decorative items, defense, and military industries. Electroplating is also becoming more and more widely used.
10 Benefits of electroplating
The deposited layer obtained by electroplating is called the plating layer, which can protect the base material from environmental corrosion, improve the appearance of the base material and increase the aesthetics.
By application, electroplating applications layers can be divided into three categories: protective, protective decorative, and functional. Among them, functional plating layers include wear-resistant, friction-reducing, conductive and optical layers, which are suitable for various fields. According to the needs of the industry served, the corresponding plating layer is selected.

(1) Formation of protective barrier
A plating layer is formed on the workpiece substrate, which acts as a barrier against corrosion. It can make the parts last longer, extend the duration of use and reduce the replacement rate.
(2) Enhances the appearance
Products coated with a plating layer look more shiny and attractive. Especially for jewelry, manufacturers can use electroplating as a low-cost method to improve the overall quality of the product and enhance competitiveness.
(3) Reduce friction
Such as electroplating nickel-molybdenum disulfide composite coating, molybdenum disulfide has a lubricating effect, mechanical intercalation in the nickel coating, not only can reduce the friction factor of the friction sub, and can effectively prevent the high load generated by adhesive wear, with good friction reduction performance.
(4) Enhancement of electrical conductivity
Silver plating is a highly efficient process for manufacturing electronic and electrical components because it enhances the conductivity of the product and is a cost-effective solution for electrical conductivity.
(5) Absorption of excess hydrogen gas
It can absorb excess hydrogen gas that is usually generated when manufacturing catalytic converters for automobiles. This can improve the performance of catalytic converters.
(6) Prevents whisker formation
Zinc whiskers originate from stresses in the coating deposit, which are generated by internal or external forces. External stresses may originate from locations such as post-processing of the product, repetitive bending, or thermal expansion.
Zinc-nickel alloy coatings can be applied to the metal parts of raised floor components during the manufacturing process, and these coatings are considered “zinc whisker free”, zinc-nickel alloys, which can prevent the formation of whisker spikes from occurring during certain types of manufacturing operations, and can also significantly reduce the risk of arcing and short-circuiting in electrical components and assemblies caused by whiskers breaking away from the material.
The damage is caused by arcing and short-circuiting components due to whisker detachment.
(7) Thermal resistance
Electroplating has good heat resistance, such as gold plated or galvanized nickel can withstand extremely high temperatures. The use of these metal coatings can protect engine components from extreme temperature damage, thereby extending their service life.
(8) Generating magnetism
The most common reason for applying plating or coating to magnets is to prevent corrosion. If your application is used outdoors, exposed to the elements, or in a humid environment, corrosion is inevitable with untreated magnets. Electroless nickel plating is often used in magnetic applications, such as the manufacture of computer hard drives. Magnetism makes the disk easier to read.
(9) Increase hardness
Certain alloy materials are brittle and prone to breakage due to mechanical stress and impact. Metal plating or conformal coating adds a layer of protection to parts, making brittle materials more durable and strong. Parts plated surface is less susceptible to damage from impacts or drops, which can extend service life.
(10) Absorption of light and energy
Black chemical nickel plating absorbs the light and energy necessary for many manufacturing processes in the aerospace, automotive and aviation industries. It ensures compliance with Department of Defense guidelines and other industry standards.

summary
The quality of the basic raw materials used for copper electroplating production is directly related to the quality of the product and the production cost of electroplating. How to control the quality and cost of these raw materials has a lot to do with the selection of suppliers.
Therefore, selecting suppliers is also an important task to be done in electroplating and barrel plating production management. Determining the best manufacturing option is critical to your company’s efficiency. Electroplating is a functionally and economically beneficial option for a variety of applications, but you need to work with the right plating company to see all the benefits. The rack plating, electroless plating, and chromium plating also help to increase production level of manufacturing companies.





