- July 8, 2022
Choosing a metal for your products is one of the biggest decisions for your project. Your choice of metal can affect the durability, costs, and overall success of the project. Galvanized metal is one of the most popular and versatile metal surface finishes due to its increased strength, durability, and formability.
In this article, we will give a comprehensive overview of the processes of galvanizing metal. Then, we will dive into the various benefits and applications of galvanized metal sheets.
Definition of Galvanizing
Galvanizing is a coating of zinc on the surface of a metal, alloy, or other material. It is a surface treatment technique that serves aesthetic and rust prevention purposes. Since zinc acts as a sacrificial coating, it protects the substrate underneath.
Therefore, galvanization is usually used for extending the lifespan of metal parts. The versatility of this metal coating makes it suitable for a wide range of applications in different industries, including automotive, solar, electronics, and construction.
What is Galvanized Metal?
Galvanized metal is simply metal that has undergone the galvanization process. The process involves applying thin zinc coatings on the metal surface to protect it from corrosive elements. It generally entails immersing the clean, oxide-free metal into molten zinc to create a zinc coating on the metal.
The coating then metallurgically bonds to the metal’s surface, acting as a sacrificial anode. As a result, the coating confers durability and longevity to the metal.

What Types of Metals Can Be Galvanized?
The most common metals that are suitable for galvanization are steel and iron. However, other types of metal can also be galvanized. Generally, ferrous metals like cast steel, cast iron, malleable iron, hot-rolled steel, and cold-rolled steel can all receive this treatment.
On the other hand, many other materials will not develop galvanized coatings. Since iron is important for the galvanization reaction, metals like copper or aluminum will not develop galvanized coatings. Materials like chrome-moly steels may develop a coating, but there will be many bare areas.
Galvanization Process: How Does Galvanizing Protect the Base Metal?
Galvanization involves adding a layer of zinc to the surface of metals like steel and iron. There are various methods of galvanizing. Therefore, the process varies depending on the technique chosen. However, they all have a general principle of surrounding the steel or iron with zinc, which could be in liquid or dust-like form.

Upon the introduction of zinc, the iron present in the metal substrate will react with the zinc to create tightly bonded alloy coatings. It is a relatively straightforward coating that makes a fairly thick coating. The complete process can be broken into three phases:
- Preparation of the metal surface
- The actual galvanizing
- Post-galvanizing surface treatment
Generally, galvanization protects the metal based on the following principles:
- The zinc coating protects the base metal from corrosive substances like acids, caustics & alkalis, gas, etc.
- Zinc acts as a sacrificial metal. Upon scratching the coating, zinc readily sacrifices its anode than the base metal, thereby protecting it from rusting.
- The ability of zinc to corrode faster than the base metal is an excellent way of protecting the metal. Some manufacturers add chromate to the galvanized metal as a “special agent” to make the zinc rust even faster.
However, preparation and post-treatment methods will determine how effective a galvanizing process will be. Insufficient preparation will affect the reaction between the base metal and the molten steel. Also, inadequate post-treatment may compromise the eventual appearance of the galvanized coating. Thus, affecting the overall quality of the product.
Different Methods of Galvanizing
There are various methods of galvanizing, with each type having its unique features and advantages. Here’s a look at them:

Hot-Dip Galvanizing
As the name suggests, this method is carried out by dipping the base metal in a molten pool of zinc. Before the actual galvanizing, the machinists must clean the base metal chemically, mechanically, or both. The cleaning is a preparation process to ensure a quality bond between the metal and the zinc coating. After cleaning, the metal is fluxed to remove any residual oxides.
The dipping process then follows in a pool of heated zinc, often maintained at a temperature of about 460°C (860°F). A metallurgical bond then begins between the zinc and the metal substrate. After pulling the metal from the bath, it reacts with oxygen to form zinc oxide. This zinc oxide also reacts with carbon dioxide to form zinc carbonate, which is the final protective coating.
Hot-dip galvanization is a quick and economical method, and you can perform it on simple and complex sheet metal designs. However, you may notice some inconsistent coatings on the metal.

Pre-galvanizing
The pre-galvanizing process is similar to hot-dip galvanizing. However, it is carried out in the first stage of production (at the steel mill). This process involves rolling metal sheets through a mechanical or chemical cleaning agent to prepare the metal for galvanizing. After cleaning, the machinist passes the base metal through a pool of molten zinc before recoiling it immediately.
This process ensures the rapid galvanization of large coils of steel sheets, creating a more uniform coating. However, some uncoated and exposed areas may become present when the fabrication of the pre-galvanized metal starts. For example, edges are left exposed when longer coils of sheet metal are cut into smaller pieces.
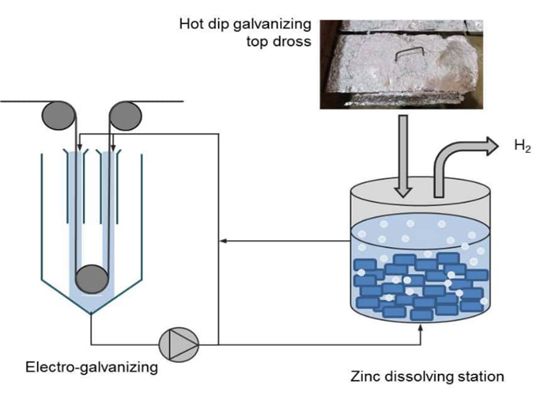
Electrogalvanizing
Unlike the other two processes discussed above, electrogalvanizing does not involve using a molten zinc bath. Instead, it involves the introduction of an electric current into an electrolyte solution before applying it to the steel material. The electric current works to transfer the zinc ions onto the metal substrate.
This method of galvanizing metal involves the electrical reduction of positively charged zinc ions to zinc metal. The zinc metal is then deposited on positively charged metal (steel or iron). Manufacturers also add grain refiners to ensure smooth zinc coatings on the metal. Like pre-galvanizing, electrogalvanizing is also applied on long rolls of sheet metal at the first stages of the product development process.
This process confers a precise and uniform coating thickness on the metal. However, coatings from this method are typically thinner than those of hot-dip galvanization.
Galvannealing
Galvannnealing is a combination of the hot-dip galvanizing and annealing processes. This process aims to create galvanized steel with a specialized coating. The annealing and hot-dipping processes occur instantaneously to produce a matte gray finish.
The hot-dipping process normally occurs before passing the coated metal through air knives. The air knives help remove excess zinc from the metal. This is then followed by brief heating of the metal in an annealing furnace at 500 to 565 °C. The zinc and iron layers then diffuse into each other, creating laters of zin-iron alloy.
The galvanized steel produced through this method can withstand welding well, and the surfaces ensure excellent paint adhesion.
Benefits of Galvanizing Sheet Metals
Overall, galvanizing confers corrosion-resistant characteristics on the metal substrate. This, in turn, offers several benefits to sheet metal. Here are some of the advantages of galvanizing metal:

1. Improve Durability
Galvanization delivers one of the most long-lasting coatings in the industry today. Industrial galvanized steel can last over 50 years in an average environment. Likewise, it can last more than 20 years with severe exposure to water. In fact, you can expect your steel structure to last up to 100 years if you use an average coating of 85 microns thickness.
The process creates a coating thickness that helps the metal meet corrosion performance requirements in today’s world. In most cases, no maintenance is required, increasing the reliability of the product.
2. Offers Three-Way Protection of Base Metal
Galvanizing protects your metal in three ways:
- It provides a physical barrier. This barrier is metallurgically bonded to the metal, preventing it from atmospheric conditions. The highly durable coating offers an adhesive strength that is over seven times stronger than organic paints. Therefore, you can be sure of your metal lasting for a longer period without maintenance.
- It provides sacrificial protection. The zinc sacrifices its anode whenever there are small scratches on the galvanized metal. The zinc will preferentially corrode in place of the underlying metal, thus, offering long-term protection.
- It prevents sideways creep. Sideways creep is a form of corrosion that occurs when rust deposits accumulate on exposed steel. Suppose there is an unusual case when the galvanized coating is compromised, exposing the bare steel. In that case, the zinc coating limits the damage to prevent this kind of damage.
3. Gets Complete Coverage
Asides from offering superior strength and durability, galvanizing also delivers superior coverage. Thus, it ensures that even the most vulnerable areas of metals stay protected for a long period. The initial cleaning process prepares and primes the metal surface for the coating. It makes the zinc coat adhere better to the metal.
Furthermore, hot-dip galvanization also allows the coating of the internal surfaces of hollow components. As a result, there’s an inside-out protection of the structure. Unlike spraying and brushing finishes, galvanized coatings will also build up at vital edges and corners.
Therefore, both small and large components can get equal levels of corrosion protection.
4. Reliable Coating
The iron elements in steel and iron alloys are prone to rust. However, combining zinc and iron element helps to create a protective layer between the metal and moisture or oxygen. The metallurgical bond formed gives secure and complete protection from the get-go. Even recesses and sharp corners could be protected with this coating, making them resistant to damage.

5. Cost-Effectiveness
Unlike many other metal treatment processes, galvanizing metal is immediately ready to use. The high-performance coating delivered by this process eliminated the need for any further parts finishing process. Furthermore, you won’t have to worry about maintenance in most cases. All these combine to reduce the overall cost of galvanized metal.
6. Fast Turnaround
Once you have a prepared metal and molten zinc ready, galvanizing takes only a few minutes to complete. Although metal preparation must be thorough, it doesn’t need to be too time-taking. Provided you work with the right company, the process will be seamless and time-efficient. Getting your products ready within a short period will help you achieve business goals faster.
Industry Applications of Galvanized Metal
Galvanized metal has a wide range of applications ranging from bodies of automobiles to drinking water pipes, nuts and bolts, electric cables, etc. Its widespread use is due to its cost-effectiveness and its ability to create durable coats. It is one of the best choices if your component will be exposed to harsh environmental conditions.
The following are some of the industries where galvanized sheet metal is valuable:

Wind & Solar Industries
Once installed, solar projects must operate continuously, and any repairs or upkeep will cost money. As a result of their ability to prevent corrosion, galvanized steels are widely used in solar installations. Because it does not emit emissions and guarantees decades without maintenance, it is also well-known for being “environmentally” friendly. Even when exposed to the harsh climate of solar projects, hot-dip galvanized steel offers corrosion protection that typically lasts for decades.

Automotive Industry
Zinc-coated car bodies are now standard in auto manufacture, although the manufacturer restricts it to high-end cars in the past. About 80% of a car’s body is its “body-in-white,” which is made entirely of galvanized steel. Galvanized steel’s ability to resist rusting makes it a useful tool for automotive prototyping since it allows businesses to offer consumers “anti-rust warranties.”
Telecommunications Industry
Due to their height and frequent difficulty in access, phone lines are quite challenging to maintain. For success, they need the strongest and most dependable materials. Phone wire and equipment boxes can be made of galvanized metal. Consequently, it lowers the danger of damage and eliminates the need for any maintenance.

Construction Industry
Galvanized steel has been a staple in the building business for more than a century, whether for commercial or residential use. In addition to its durability, it is also used for construction due to its aesthetic appeal. Galvanized steel has a contemporary feel, thanks to its “shine,” and designers frequently use it in modern architectural design concepts.
Additionally, people use it for a variety of smaller structural components as well, including fencing, rails, gutters, tubes, poles, and more.
Conclusion
Galvanized metal offers several benefits, including durability, corrosion resistance, and cost-effectiveness. This type of metal is useful for a wide range of applications in many industries. Whenever your applications require products that will be exposed to intense environments, galvanized sheet metal should be your option.
If you are unsure about the type of material or surface finishing to choose for your product, let our experts at WayKen help you. We boast in-depth knowledge of surface finishing and quality manufacturing services. A perfect combination of expert technicians and state-of-the-art technology helps us deliver outstanding services. Contact us today for your metal fabrication, and let’s bring your concept to life.
FAQs
How can I tell if a metal is galvanized?
The pattern on the metal surface will tell you if it is galvanized. Galvanized metal often shows a distinctive crystalline-like pattern known as the “spangle” pattern. Since the process also bonds a zinc layer onto the metal, you can also check by using thickness measurement, laboratory testing, and electron paramagnetic resonance (EPR).
Which is better, aluminum or galvanized steel?
Galvanized steel is preferred to aluminum for industrial processes where components will be exposed to corrosive materials because it offers better corrosion resistance than aluminum. However, aluminum products are preferred when applications require lightweight materials and efficient heat conduction.
Can you etch galvanized metal?
Yes, it is possible to etch galvanized steel. To achieve this, you must remove as much of the zinc layer as possible using fine sandpaper or a mildly abrasive pad. The acid etches will then reach the steel and etch the desired design on the metal.





