- January 6, 2023
Manufacturers use different metal plating finishes for custom products. Most of these metal plating surface finishes improve the aesthetics of products, while others enhance functionality, and some offer both.
Metal plating remains a practical choice for post-production finishing and a significant aspect of modern technology, making it an excellent choice for many applications. This article explores the metal plating surface finish and its principle, detailing the essential elements of this technique and how you can get the best from it.
What is Metal Plating Finish?
Metal plating finish involves the creation of an outer layer on a component using chromium, copper, nickel, or any other metal to improve its aesthetic and resistance to corrosion. A plating finish offers numerous benefits to parts made from metal and other materials.
It helps improve machined parts’ surface roughness. This finishing technique provides an aesthetic appeal, reduced friction, improved hardness, improved paint adhesion, and solderability.
Working Steps for Metal Plating Process
The metal plating process comprises several steps from pretreatment to post-treatment. There are several plating techniques; however, this aspect will focus on the basic procedures of electroplating:
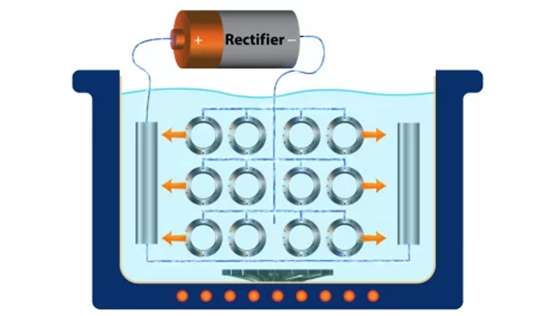
Step 1: Pretreatment
The pretreatment helps to remove all harmful contaminants on the substrate that may hinder the plating process. It may involve cleaning, rinsing, and other pretreatments that rid the plating surface of contaminants.
Step 2: Determine the Effectiveness of the Cleaning Process
The requirements of your parts often decide the ideal plating process to use. Likewise, it determines how clean your substrate must be before plating occurs. For instance, some CAD plating techniques only require the removal of bulk soil and dirt, while others require the removal of oil and grease.
Step 3: Set up the Plating Station
As electroplating is the focus here, you need materials such as an anode, a cathode, a rectifier, a tank, and a suitable plating solution to set up your plating station.
Step 4: Plating process
The deposition process starts as soon as you turn on the electrical current. In cases where a thicker plated finish is required, you must expose the components to the current for longer. However, you may have to consider variables such as temperature, voltage levels, immersion durations, etc.
Step 5: Post-Treatment Process
Post-treatment cleaning is necessary after achieving the desired deposition on your product. Therefore, electrolytic polishing is an effective means of improving the corrosion resistance of manufactured components.
Step 6: Waste Disposal
The metal plating finish produces heavy metals, which are usually hazardous because of their high toxicity levels. As a result, an efficient way to dispose of this waste is by pretreating the plating wastewater before its disposal.
Different Types of Metal Plating Finishes
Various standard industrial plating finishes are available for use. Here are some of them:

1. Zinc Plating
Zinc remains one of the most cost-effective materials that create galvanized metal on metal substrates. Manufacturers use zinc through molten bath dipping and spraying. The substrate takes the cathode position while the metallic zinc takes the place of the anode in a soluble zinc salt electrolytic bath. The zinc plating technique forms a very ductile coating on plated metal, giving you better control over thickness and uniformity.
2. Chrome Plating
Chrome plating or alodine finish generally involves using chromic acid and trivalent chromium baths to create an overlay on the metal components. The primary purpose of chrome plating is to enhance the aesthetics of the material.
However, chromium plating finishes increase the hardness of the material, thereby giving it a better corrosion resistance property. Chrome plating can restore tolerances on worn-out parts. These chrome-plated components are generally suitable for industrial applications.

3. Nickel Plating
Nickel is another common plating metal valuable in the electroless plating method. Nickel plating helps to create a protective coating on household products such as doorknobs, shower fixtures, cutleries, etc., and it helps to improve its aesthetics and wear resistance properties. This plating method is suitable for aluminum and copper. However, it works well on other metals, serving as a basic plating for chromium.

4. Copper Plating
Copper is the most cost-efficient metal plating that offers high conductivity. Copper plating is standard for electronic products such as circuit boards, and it is a common choice because it provides high plating efficiency and low material cost.

5. Cadmium Plating
Cadmium plating is a metallic silver tender coating deposited on different base metals such as aluminum, steel, copper, and iron. This process is suitable for coating bolts and screws for submarines and military parachute buckles. In addition, it has high malleability, corrosion resistance, ease of solderability, and chemical resistance.

6. Silver Plating
Silver plating is identical to gold plating as it improves the aesthetic appeal of the material used. It is an excellent metal finish type when electrical conductivity is a factor you need to consider. Many manufacturers use silver plating because of its cost-effectiveness, as it is a better alternative to gold. Also, it is effective for plating parts made from copper.

7. Gold Plating
This metal is famous for its resistance to oxidation and its high electrical conductivity. Gold plating is an effective means of transferring these remarkable properties to metals like copper and silver. It has an enormous application in enhancing the conductivity of electronic parts like electrical connectors.

8. Tin Plating
This industrial finish involves depositing a solderable tin layer coating on a material’s surface using electrical current. Electroplating tin is a very cost-effective process because tin is readily available and costs less than metals like gold, palladium, or platinum.

9. Rhodium Plating
Rhodium plating is another standard metal plating finish that deposits a thin coating of rhodium on the surface of a piece of jewelry. This process is the same as rhodium dip or rhodium flashing. It helps to enhance the luster, durability, and light reflection of a jewelry piece.
Metal Plating Techniques for Custom Products
There are various metal plating techniques used in different industries. Here are some of them:
Electroplating
Electroplating is a metal plating technique that involves depositing a metal or metal alloy on the surface of a workpiece. It is a typical metal plating process involving electric currents in an electrolyte solution to dissolve charged metal particles (ions).
The charged (positive) metal ions get attached to the workpiece, while the workpiece, in this case, possesses a negative charge on the circuit. The dissolved metal particles rise to the material’s surface when you put the workpiece in the chemical solution.
Electroplating enhances the chemical, mechanical, and physical properties of a workpiece. It provides a smooth, quick, and even coating on the plated metal. This thin coating forms a protective and aesthetic appearance on the metallic workpiece.
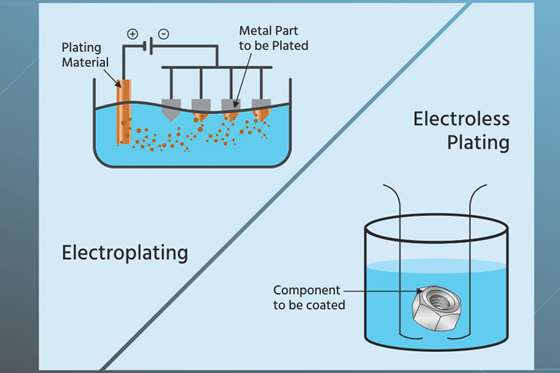
Electroless (Autocatalytic) Plating
The electroless plating technique is the ideal alternative to electroplating and is a much simpler and cost-effective option. It is also known as autocatalytic or chemical plating because it does not require any external electric current.
Electroless plating deposits metals such as copper, gold, nickel, and silver on the surface of different materials using a reducing chemical bath. It produces catalytic reduction to the path because it is a chemical process that does not require machinery or electricity.
Electroless plating is suitable for hard surface components with excellent corrosion resistance. As a result, the oil and marine industries utilize this process in manufacturing their components. Some common components, such as pumps or valves, often exposed to corrosion, are created with electroless plating technology.
Immersion Plating
The immersion metal plating process involves immersing a metal into a chemical compound that contains metal ions of a noble metal. The ion present in the noble metal usually is stable.
Thus, a natural pull sets in to dislocate the metal ions from the initial metal giving it a thin layer of noble metal ions. This process is a bit slower than electroless and electroplating processes. Also, it is only suitable for noble metals like platinum, silver, and gold.
Carburizing
Carburizing is a metal plating process that solidifies the surface of a metal while allowing the metal beneath to remain soft. This technique is the same as carburization, surface, or case hardening, and it results in the formation of a thinner and solid layer of metal called the surface case.
Manufacturers carburize a part after it has been machined to its final form as it helps to make element contents harder, as in pattern welding. Carburizing is an ideal metal plating technique product designers use to prevent corrosion in industries that use metals.
Physical Vapor Deposition (PVD)
This type of metal plating is a thin film preparation method that physically sublimes the surface of a material source (liquid or solid) gaseous atoms, molecules, or into ionized into ions under vacuum conditions. A film with a specific function is then deposited on the surface of the substrate using a low-pressure plasma or gas.
The essential physical vapor deposition technologies are sputtering deposition, ion plating, vacuum evaporation, arc plasma plating, etc. PVD coating has rapid deposition, strong adhesion, excellent diffraction, and an extensive application range.
Plasma Spray Coating
This metal plating is a highly flexible and effective thermal spray technique that involves spraying heat-softened or melted materials on the surface of a material to achieve the preferred coating. It uses a powerful spray coating technology that can enable the spraying of various materials such as ceramic, metal, and cermet.
Plasma spray applications include jet engine protection, restoring worn parts, and more. The plasma spraying process offers excellent resistance to wear, abrasion, electricity, heat, and oxidation.
Advantages and Disadvantages of Metal Plating

Metal plating surface finishes have many benefits, which attract several industries. The common benefits of metal plating are:
- It increases the aesthetic appeal of the material used to make a component.
- The different metal plating finishes are compatible with an extensive list of materials.
- It helps to protect components from wear and corrosion.
- Metal plating is a cost-effective finishing method.
- It enhances the hardness and strength of a material.
Although this post-processing technique has a wide range of benefits for manufactured parts, it also has some drawbacks, including:
- The plating can chip and crack in specific environments due to wear.
- The metal plating process can be lengthy, compromising a component’s specification.
Applications of Metal Plating Finishes
Several industries adopt varying metal plating finishes to the best of their product specifications. These finishes are ideal options for different industries. Here are some of their primary applications:
Aerospace
The aerospace sector electroplates its components to achieve a sacrificial coating. Generally, it increases the path lifespan by minimizing corrosion. Adding an extra metal layer to parts can enhance functionality. This is because aircraft components are usually exposed to considerable temperature changes and several other environmental elements.
Automotive
Automakers commonly use electroplating to create customized parts for concept vehicles. Various vehicle customization businesses and restoration industries utilize electroplating in applying nickel, chrome, and several other finishes for motorcycle and vehicle components. In addition, vehicle makers use black oxide coating as it is ideal for machined parts and can last longer.
Power
The power industry electroplates solar components to enhance their electrical conductivity. Manufacturers electroplate solar cell contacts and antennas. Also, they electroplate wires using silver, nickel, and other metals.
Dental and Medical
The medical sector often uses metal plating to add toughness to the components’ surfaces in all medical and dental instruments, preventing metal deterioration. Manufacturers produce tooth inlays with gold plating making them helpful for various dental procedures.
Jewelry
Electroplating is one of the most widely used techniques in the jewelry sector. Manufacturers and designers often use this method to enhance the aesthetic appeal, durability, and color of bracelets, pendants, rings, and other accessories.
Factors to Consider for Metal Plating Surface Finishes
Metal surface finish, if done right, offers an extensive range of benefits ranging from durability, functionality, and aesthetics. As a result, to get the best results from metal plating finishes, here are some of the critical that determine how to plate metal:
Pre-plate Material Conditions
The material must be in a plating-ready condition before you can plate such material. For instance, if a substrate has heat treatment scales, oxides, oil buildups, and other residues, the plated deposit may fail to stick to the surface of such substrate.
Therefore, you must remove these buildups with pretreatment procedures such as acid pickles, vapor blasting, chemical descalers, deoxidizers, alkaline presoaks, etc. However, a bead blast finish is an effective means of preparing a plating surface.
Thickness Tolerance
Plating complications may occur due to excess or low coating thickness. The standard reality is to decide the adequate range of the preferred maximum and minimum thickness. Nevertheless, here are some key factors to keep in mind to make the range achievable and specific in your metal plating finishes:
- Electroplating creates even coating in most cases. As a result, tight thickness tolerances will not be a problem.
- Mark out a workpiece’s surface areas that must be within a tight metal plating thickness. It is vital to consider recesses, corners, and other geometric parameters.
- Electrolytic plating makes finishes settle on some regions of a plating surface more than others. For example, plating deposits become thinner within ¾ inches of a corner or recessed area. Consequentially, it may be unadvisable to use a tight range tolerance for flat and simple shapes.
- It is essential to create checkpoints to decide the plating tolerance requirements. Ensure you have such checkpoints on the surfaces where it is most suitable.
Thread Inclusion
Examining the other thickness aspects for workpieces that feature any machining thread is essential. Plating workpieces, such as hydraulic fittings, screws, etc., are typically about four times thicker than flat surfaces. Thus, it would help to consider the buildup into your desired plating process. It helps to ensure that the threaded workpiece meets the essential specifications and fits perfectly.
WayKen Metal Plating Services for Custom Products
It pays to partner with experts to achieve the best results in your machining projects. Therefore, as you consider metal plating finishes for your applications, WayKen is the right partner. Our parts finishing services provide the best possible option for your custom projects.
WayKen has good years of experience providing cost-effective metal finishing services for an extensive range of industries. There are several metal plating options to choose from to improve the functionality and durability of your components. Our professionals work with you to provide economical solutions to satisfy your metal finishing needs. Contact us today, and let’s get started with your project!
Conclusion
Metal plating surface finishes are crucial in ensuring that your products have excellent aesthetic appeal, resistance to wear and tear enhanced longevity, and are highly resistant to corrosion. However, not knowing what your products need can be a significant constraint. As a result, you need to understand your project needs and identify the ideal finishing option.
FAQ
Does gold plating wear off?
Metal plating, such as gold plating, can wear off or tarnish over time, losing its brightness. However, note that wear and tear is a common reaction that occurs irrespective of the component’s quality.
Can metal plating prevent rust?
The plating technique maintains the permanent safety of machines and several tools from rust and corrosion. This is because the thin layer deposited on the surface of these tools helps to keep their durability and resistance to corrosion.





