- September 2, 2022
Stainless steel is a universal material that forms the foundation of innumerable industrial setups. Passivation of stainless steel is a core process in maintaining its quality and longevity.
In this article, we dive into the important questions of how to passivate stainless steel and what is it.
What is Stainless Steel Passivation?
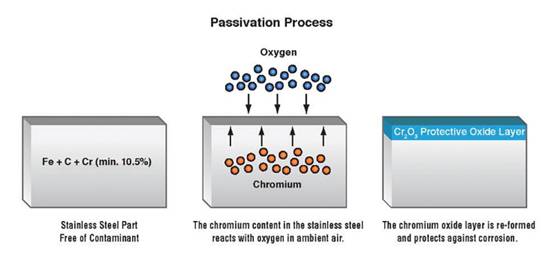
Passivation of stainless steel is a chemical process to make it unreactive to moisture and oxygen, the primary substances that cause it to rust. The passivation process achieves this by creating a very thin layer (a few atoms wide) of Chromium Oxide (2Cr2O3) on the stainless steel’s exposed surface.

This layer prevents atmospheric compounds from reacting with the iron content in stainless steel, which deteriorates its quality. The results are quite similar to aluminum anodizing.
It serves a very important purpose in numerous industries like food processing, cosmetics, pharmaceuticals, etc. where equipment corrosion can lead to safety hazards and great losses. Properly passivated stainless steel also improves productivity and decreases maintenance load.
Why Passivated Stainless Steel is Needed?
The thin layer of chromium (and nickel) oxide on stainless steel makes it resistant to corrosion. However, sometimes this layer may not properly form by itself or may be damaged, leaving the stainless steel susceptible to corrosion. This is where the passivation process comes in. It serves the purpose of forming or restoring the protective layer to the desired quality.
There are several reasons stainless steel may require passivation, some of which are as follows:
–Low-Quality Raw Materials: Impurities from bad quality raw materials may not be fully extracted during steel fabrication. This can cause its chemical passivity to fall.

–Machining/Grinding: Most stainless steel parts go through manufacturing processes like machining and grinding. The tools used in these processes contain iron that gets embedded into the stainless steel surface during machining. Machining lubricants and cooling liquids can also accumulate in surface pits, introducing another form of impurity.
–Welding: Welding weakens the chromium oxide layer, especially in the heat-affected zone.
–Assembly: Stainless steel products are most often an assembly of various parts. During assembly, individual components rub against each other and the resulting friction can damage the layer of oxides, oftentimes to the extent that it may completely vanish at some points.
–Regular Wear & Tear: The chromium oxide layer weakens over time as the part sees various thermal, chemical, and physical environments during service.
All of these scenarios are very common in an industrial environment. Thus, professional engineers adopt a regular steel passivation schedule to prolong the life and quality of industrial equipment, saving money, time, and effort in the long term.
3 Steps for Stainless Steel Passivation Process
We will now discuss the passivation process in depth. Stainless steel passivation is a 3-step process: cleaning, acid bath, and quality testing.
Step 1: Cleaning
Cleaning the stainless steel surface is a crucial step for successful passivation. Accumulated surface impurities can block the passivating acids from reaching the surface, rendering all efforts useful.
Common surface impurities include dirt, grease, mineral and synthetic oils, hydrocarbons, and other residues from industrial environments.
There are various methods to degrease a stainless steel surface. Alkaline detergents and high-temperature baths (up to 65°C) are highly effective in dissolving and removing impurities.
Oftentimes, engineers also check the quality of cleaned surfaces via techniques like the camphor test.
Step 2: Acid Bath
The second step for the passivation of stainless steel is the acid bath. The cleaned stainless steel surface is immersed in a bath comprising an acid solution and some other additives like accelerators and inhibitors.
This chemical reaction removes free iron from the metal’s surface. The main goal is to achieve a high chromium-to-iron ratio so there is more chromium available for oxidation instead of iron.
Three parameters control this step in the passivation of stainless steel: the concentration, temperature, and duration of the acid bath. Different combinations of these parameters yield different results. The engineers’ experience and skill are critical in setting these parameters for the best results.
The most common acids for steel passivation are nitric acid and citric acid. In the following section, we briefly touch upon both of these and offer a comparison.

Nitric Acid vs. Citric Acid: A Technical Comparison
Nitric acid was the original passivating agent for stainless steel that works on the metal by dissolving iron, which the circulating bath then carries away. Its main features are:
- The concentration of nitric acid solution ranges from 10%-50%.
- Acid bath temperatures can be up to 60°C.
- Additives like sodium dichromate and phosphoric can further improve its passivation efficiency.
- Nitric acid makes passivated stainless steel more resistant to flash attacks.
- Only applicable to austenitic and duplex steel grades.
- Highly toxic and dangerous for the environment, requiring careful handling and disposal.
Let us move on to citric acid. It is a relatively new agent for the passivation of stainless steel. It chemically reacts with iron on the steel’s surface but also removes chromium and nickel. However, the prepared surface is well prepared to naturally react with ambient air to form a thick oxide layer.
- Acid concentration of around 10% is used.
- Passivates all grades of stainless steel.
- Citric acid is safe to use and is relatively more environmentally friendly.
- Approved for food processing.
- Passivation by citric acid is quicker and can be adjusted to achieve results in as less than 5 minutes.
Step 3: Quality Testing
The last step in stainless steel passivation is quality testing. The passivated metal passes through some checks that it needs to pass.
Engineers use several testing methods at this stage. One strategy is to create an environment where rusting is easy. This is known as the water immersion test, where stainless steel goes through continuous cycles of wet and dry environments and is constantly monitored for signs of corrosion. Usually, poor passivation leads to visible rust on the surface.
Chemical tests like the Ferroxyl test are also quite useful. It exposes iron by reacting with it and changing its appearance. Also, equipment like passivity meters is also quite popular to check the reactivity of stainless steel.
Meanwhile, it is also common to have quality checks during the acid bath step. Monitoring the iron concentration and acidity of the circulating acid bath indicates the progress and effectiveness of the passivation process.
Common Industry Standards for Passivation of Stainless Steel
Passivation of stainless steel is highly standardized due to its importance. Industry standards by ASTM and ASM are the most popular ones, covering passivation of useful stainless steel grades like 304 and 316 SS.
ASTM
The American Society for Testing and Materials (ASTM) has two comprehensive standards for stainless steel passivation.
The ASTM A967/A967M standard covers various types of chemical passivation with both nitric and citric acid, as well as electrochemical treatment. It contains detailed information on the passivation process, quality testing methods and criteria, and recommendations for the initial steps of cleaning and descaling.
The ASTM A380/A380M contains detailed process guidelines and precautionary statements for passivating stainless steel parts, assemblies, equipment, and installed systems. This includes various steps of the process like cleaning, descaling, and passivating.
AMS
SAE International maintains the AMS 2700 standard detailing specifications for passivating stainless steel surfaces. It is more tuned towards professional practices in industries like aerospace and automotive.
It provides guidelines and procedures for dissolving iron and other less noble metallic elements from the surface of stainless steel to make it more corrosion-resistant.
Equipment Features for Stainless Steel Passivation
There is a wide range of machinery for the passivation of stainless steel. We will highlight some of the main features of the passivation equipment available in the market.

Size: There are several sizes of passivation equipment from benchtops to large-scale industrial units that can passivate huge parts.
Passivating Capacity: This is defined by factors like the number of tanks, their volumes, and overall throughput.
Functionality: Some passivation equipment comes as a complete package for degreasing, passivating, rinsing, and drying.
Manual/Automatic: Users can choose between manual and automatic units. Automatic units are more accurate, safe, and have intuitive human-machine interfaces. PLC-controlled systems add another level of precision and reliability to the machine.
Good Practices for Passivated Stainless Steel Parts
As with any industrial procedure, certain professional practices enhance the quality of steel passivation processes. Enlisted below are some of the major recommended practices for engineers who deal with passivated stainless steel.

1. Always Passivate New & Repaired/Replaced Parts
Every time a new component is added or an old one is replaced, passivation is a must. A chemically reactive part can very quickly catch rust and damage other parts and also the industrial process itself.
2. Regularly Monitor Equipment
Most industries use stainless steel parts whose passivity decreases over time with use or due to compounds like chlorides. The corrosion potential of such components must be periodically checked to see if they need re-passivation.
3. Separate Machinery & Tooling for SS
It is economically prudent to process stainless steel in a separate environment, where it may be less exposed to iron and impurities. In the long run, it can save massive passivation costs and downtime.
4. Quality Control of Industrial Fluids
An industry working with corrosive fluids should keep their composition under check. The fluids should be cleaned or changed if they become too dangerous for the stainless steel equipment.
5. Adjust Passivation Method to Steel Grade
Engineers must understand that every stainless steel grade is different and the same passivation cycle is not fit for all of them. Thus, some intuition and experience are required to tweak the process for each grade and part to achieve optimal results.
Conclusion
Stainless steel passivation is an important process that helps to keep stainless steel looking new and prevent it from corroding. So, it is inarguably a powerful method to boost productivity and quality of service.
While the process is not complicated, common passivation even can be done at home with a simple kit. If you want production-grade stainless steel passivated parts, then you will need to hire a professional company.
Choose A Reliable Post finishing Partner for Your Passivation Project
For manufacturing services ranging from part production to the passivation of stainless steel, WayKen is your right choice. With advanced technology and a rich experienced team, we can handle all your requirements and deliver results in the shortest time.
No matter stainless steel passivation to all the other surface finishing processes, you’ll confidently get parts with durability and perfect performance. Contact us for your passivation projects, and we’ll give you an instant quote and DFM feedback within the next 12 business hours.
FAQs
Does 304 vs. 316 stainless steel need to be passivated?
Passivation is recommended for both 304 and 316 grades for long service life. While 316 SS does have better corrosion resistance due to its higher molybdenum content, it does not provide enough protection for most industrial environments.
How often should you passivate stainless steel?
The frequency of passivation for stainless steel depends on your industrial application. Parts that are exposed to too much dirt, friction, wear or other extreme conditions should be passivated more frequently than parts in a mild industrial environment. Parts carrying high chloride content, for example, should be passivated 2-3 times a year. Generally, SS parts should be passivated once a year for normal applications.
Can you passivate stainless steel twice?
Yes. The protective oxide layer on stainless steel gets weaker with time and use. When this happens, stainless steel should be re-passivated to restore its corrosion resistance. Depending upon the condition of the SS parts, they may be passivated more than twice as well.





